Which CBD Absorption Method Provides The Best Bioavailability?
Best CBD Absorption Methods For Bioavailability
Cannabidiol, the non-intoxicating phytocannabinoid from the hemp (cannabis) plant, seems to be infused in everything from food and drink, to oils, balms and anything and everything that can be imagined.
With such a wide range to choose from, how do you know which one will work best for you? For many people, consideration can be based on preference, tolerability, palatability, digestibility, and familiarity to name but a few.
Possibly the most important question and unfortunately often overlooked when choosing CBD, is which product offers the greatest bioavailability?
What Is Bioavailability?
Bioavailability is simply the percentage of CBD that enters your systemic circulation (your general bloodstream) post administration.
Think of it this way, when you eat a CBD chocolate bar containing 100mg of CBD, by the time that piece of chocolate passes through your digestive system, and enters your systematic system it now resembles closer to 10mg of CBD. Bioavailability of CBD differs greatly depending on the method of administration. It can range from 100% to 6%.
So which method gives you the greatest bioavailability and ultimately the best value for money?
CBD Absorption Methods
Intravenous
The most direct method with 100% bioavailability is the intravenous method. Here, CBD is injected directly into your veins meaning the entirety of the dosage is now in full systemic circulation.
The intravenous route is the most reliable from a pharmacokinetics point of view in laboratory trials, as it reduces individual variation of bioavailability which could affect the results.
For the general public though, it is hard to imagine this intrusive and time-consuming method, albeit rapid and efficient, gaining in popularity any time soon.
Oromucosal
Pharmaceutical drug Sativex which contains both the psychoactive cannabinoid THC and the non psychoactive CBD when administered the oromucosal (via the gums/cheeks of the mouth) route, demonstrated bioavailability of 95%!
CBD spray products are available to use for the oromucosal route.
Vaping CBD
Vaping CBD is quickly becoming incredibly popular around the world as an efficient and effective method of consuming CBD. It is also incredibly safe as none of the toxins present in combustion occur in vaping due to the fact that the CBD is heated not burned.
Vaping involves heating CBD (liquid or flower) in an easy to use, portable, electronic device. The vapor is inhaled into the lungs, there, CBD enters the systemic circulation rapidly via pulmonary absorption.
Bioavailability can average anywhere between 50 – 70% but has limitations due to the fact that some CBD is lost in exhalation.
Vaping is also known as the best method to activate other cannabinoids and terpenes present in full spectrum CBD, because vaporizers can accurately control temperatures to a single degree.
Intranasal
The nose offers fast systemic absorption as the naval cavity is covered in thin mucosa that is well vascularised allowing CBD to enter the systemic blood circulation quickly without having to go through the digestive system.
Studies on accurate bioavailability in humans are non existent at present, but one study in animals showed a bioavailability between 34%-46% for CBD intranasal administration.
Sublingual
A common method of consuming CBD is sublingually. Using a CBD tincture, drops of CBD are placed under the tongue and held there for a minute or so before swallowing.
Beneath the tongue is the sublingual region which contains numerous blood vessels and nerves which allow for rapid absorption of drugs into the systemic circulation that limited research suggests provides a bioavailability of 15%-35%
READ MORE: Discover The 10 Best CBD Oil Brands In The UK For 2019
Given the proximity and similarity of Oromucosal administration, and numerous research articles on increased bioavailability of other drugs using the sublingual method, a higher bioavailability for the sublingual method is predicted
That being said, 100% bioavailability is unlikely, as salivary enzymes under the tongue risk degrading some of the CBD and a portion of CBD is naturally going to be swallowed into the digestive system.
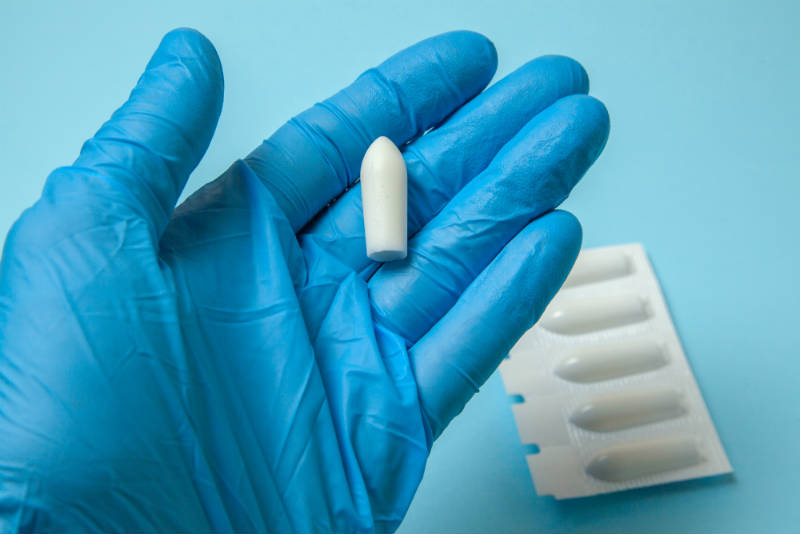
Rectal Suppositories
Using a rectal suppository, CBD is quickly absorbed into the cellular walls and mucus membranes of the anus, in particular, the lower, middle and upper haemorrhoidal veins.
The lower and middle veins allow CBD to bypass the digestive system, therefore rapidly entering the bloodstream. The upper vein on the other hand leads into the portal veins which drain into the liver leading to a loss of CBD via first pass metabolism.
Unfortunately, there are no official studies to determine the total bioavailability of rectal suppositories and estimates of anywhere between 20-60% are only made on the basis of CBD entering the lower and middle haemorrhoidal veins which is not sufficient enough to be definitive by any means.
Suppositories are readily available and are becoming more and more popular in particular for those who cannot swallow food or pills.
Orally Drink & Food & Capsules
As appealing as CBD edibles and drink may sound, the bioavailability of CBD when taken orally, like CBD capsules, can be as low as 6% according to recent studies.
When CBD is taken orally it naturally goes through the digestive system. CBD passes from the lumen of the digestive tract, through the hepatic portal vein into the liver.
READ MORE: CBD Oil Domain Registration Name Sells For $500,000
The liver is the main organ that metabolizes drugs. This breaking down action by the liver is called first pass metabolism. CBD, like many drugs, has a heavy first pass effect meaning a significant amount is removed by the liver and very little reaches systemic circulation.
Liposomes
A technique used by some drug, nutrient supplements, and CBD manufacturers is to encapsulate the desired compound in what is known as a liposome. Liposomes are spherical vesicles having an aqueous core enclosed by one or more phospholipid bilayers.
CBD is enclosed in these double layered bubbles which protect CBD from digestive enzymes and the first pass effect in the liver.
Although no specific data relating to CBD bioavailability when used with liposomes exists, there are numerous studies proving liposomes increase bioavailability in poor water-soluble compounds.
Transdermal
Topical lotions, balms and creams are highly prevalent in the market. Bioavailability scores are currently unavailable and further research is needed.
Dont forget to share this post with friends!