GW Pharma What Their Game?
Who Are GW Pharma?
GW Pharmaceuticals is a world-leading company specialising in cannabinoid (cannabis-based) medicine development. They were the first organisation in the world to obtain a license to sell a cannabis-based prescription medicine.
Doctors Geoffrey Guy and Brian Whittle founded GW Pharmaceuticals in 1998. The organisation currently has operations in the UK and the US. They work entirely in plant-based medicines using their drug discovery and development process, intellectual property expertise, and collective lab experience.
One year after their founding, GW ran their first clinical trials of cannabinoid medicines. They work closely with the UK government Home Office and MHRA the Medicines and Healthcare Regulatory Authority. Since the beginning, they claim to follow the highest standards of laboratory management and cleanliness.
What Do They Do?
CBD
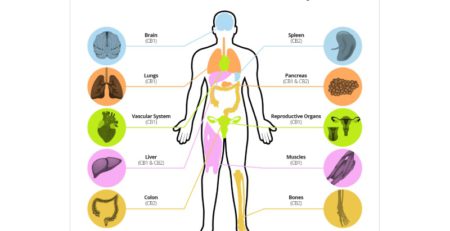
CBD stands for cannabidiol and is an oil produced by the cannabis plant. It is non-psychoactive, meaning it does not cause a high. CBD is thought to reduce neuropathic pain and spasticity in MS patients. THC, on the other hand, is psychoactive and is also thought to have great medicinal effects. Growing evidence also suggest CBD may reduce the psychoactive effect of THC.
Clinical Trials
Since obtaining the necessary licenses, GW has been working on a range of cannabis-based treatments. Both pure CBD and a CBD/THC 1:1 mixture are developed and sold by GW. These are named Epidiolex and Sativex, respectively.
Sativex alone has so far been tested in over 20 double-blind, placebo-controlled clinical trials involving over 8000 patients. This kind of trial is the gold standard in drug testing. After this, GW obtained licenses to sell their products as drugs in several countries for MS (Multiple Sclerosis)-induced pain and Dravet Syndrome a rare form of epilepsy.
How Does It Effect CBD?
GW Pharmaceuticals is one of the only private companies dedicated entirely to the research and development of medicinal cannabis products. Raphael Mechoulam research group at the Hebrew University of Jerusalem is a comparable organisation with similar time and resources dedicated to cannabinoid research.
Whether corporations like GW Pharma will positively or negatively effect CBD is down to one own opinion. Some would say that corporate marijuana is a bad thing, however, plenty of big, unscrupulous companies have thrown their weight in behind cannabinoid therapy already. Due to the exploding market, there is little to stop certain companies selling counterfeit products, and in the last three years, the FDA in the US have issued a number of letters of warning to groups selling CBD products containing no actual CBD!
Perhaps licensed, accountable groups like GW Pharma and Mechoulam team will lead to the availability of a pure and safe product that can be rigorously tested so that its benefits can be felt by everyone.